Executive Summary
Endocrinopathic laminitis is increasingly understood as a receptor-mediated structural failure of the lamellar attachment system rather than a primarily inflammatory hoof disorder. The strongest upstream driver is insulin dysregulation, particularly sustained or exaggerated hyperinsulinemia, most commonly associated with Equine Metabolic Syndrome and frequently seen in horses with Pituitary Pars Intermedia Dysfunction (PPID) overlap.
Controlled experimental work has shown that prolonged hyperinsulinemia can induce laminitis under euglycemic conditions, supporting insulin exposure as a causal trigger rather than a simple correlation. The insulin–IGF-1 receptor theory proposes that persistently high insulin concentrations stimulate Insulin-like Growth Factor 1 Receptor (IGF-1R)–linked signaling in lamellar epithelial cells, promoting proliferative remodeling, epithelial stretch, and progressive weakening of the dermo-epidermal junction.
This model integrates:
- Molecular signaling pathways
- Hoof biomechanics
- Gut–endocrine physiology
- Diagnostic interpretation
- Seasonal endocrine variation
- Field-based mechanical observations
Introduction
A common clinical scenario challenges the traditional “inflammation-first” model:
A horse presents with laminitic pain and early hoof capsule distortion—yet there is no colitis, endotoxemia, grain overload, or systemic inflammatory disease. Radiographs may initially show minimal displacement, while farriers detect subtle growth ring distortion, altered breakover, or increased sensitivity.
These cases increasingly align with endocrine-driven laminitis rather than sepsis-associated mechanisms.
Understanding the insulin–IGF-1 receptor pathway clarifies:
- Why hyperinsulinemia can precede structural failure
- Why glucose concentration alone is not the primary risk metric
- Why lamellar remodeling can occur without overt inflammation
| Feature | Endocrinopathic Laminitis | Sepsis / SIRS-Related Laminitis |
|---|---|---|
| Primary Driver | Hyperinsulinemia / insulin dysregulation | Systemic inflammation / endotoxemia |
| Typical Systemic Illness | Often absent | Often present |
| Dominant Tissue Pattern | Structural remodeling / epithelial stretch | Inflammatory mediator-driven injury |
| Practical Framing | Endocrine / metabolic pathway | Inflammatory / critical-illness pathway |
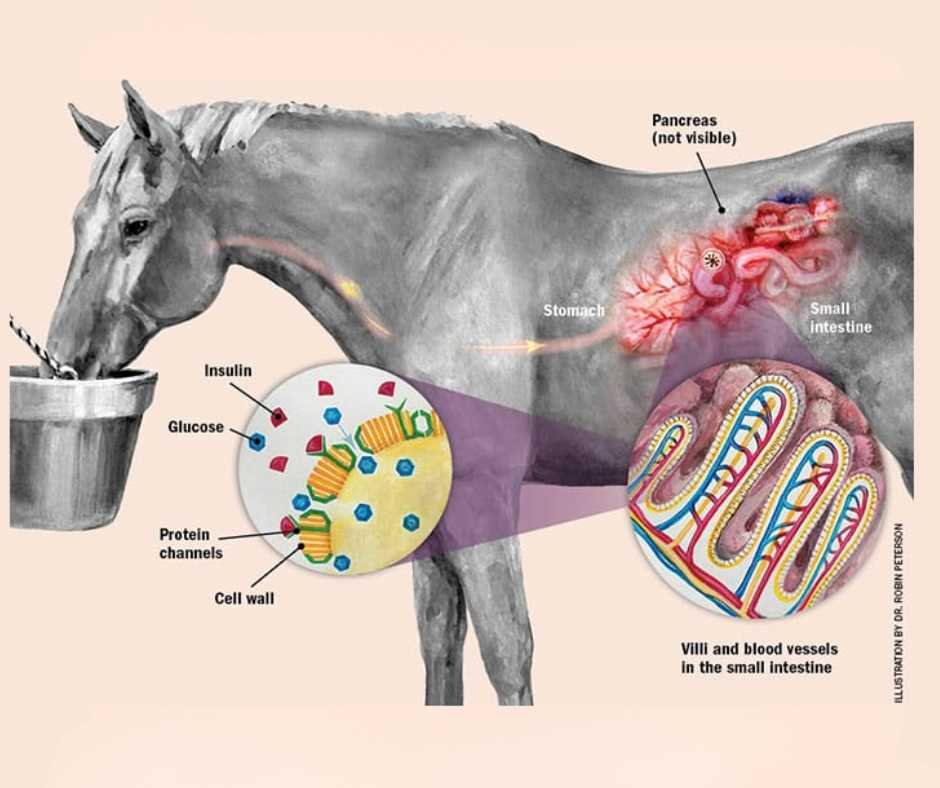
Insulin Dysregulation as the Metabolic Foundation
Insulin dysregulation includes:
- Resting hyperinsulinemia
- Exaggerated post-prandial insulin responses
- Variable insulin resistance
A landmark experimental study demonstrated laminitis induction via prolonged hyperinsulinemia in clinically normal ponies while maintaining normal glucose levels..
| Pitfall | Why It Distorts Interpretation | Practical Consequence |
|---|---|---|
| Inconsistent feeding status | Alters insulin baseline and post-feeding response | False reassurance or unnecessary alarm |
| Stress or excitement at sampling | Stress hormones can shift endocrine output | Misclassification of metabolic risk |
| Single time-point reliance | Misses post-prandial insulin peaks | Under-detection of insulin dysregulation (ID) |
| Ignoring ACTH seasonality | Seasonal ACTH rise skews reference ranges | PPID over- or under-diagnosis |
Effects on Lamellar Epithelial Cells
Lamellar epithelial cells are engineered for precision structural suspension of the distal phalanx.
When growth-oriented signaling is overstimulated:
- Secondary epidermal lamellae stretch
- Attachment complexes weaken
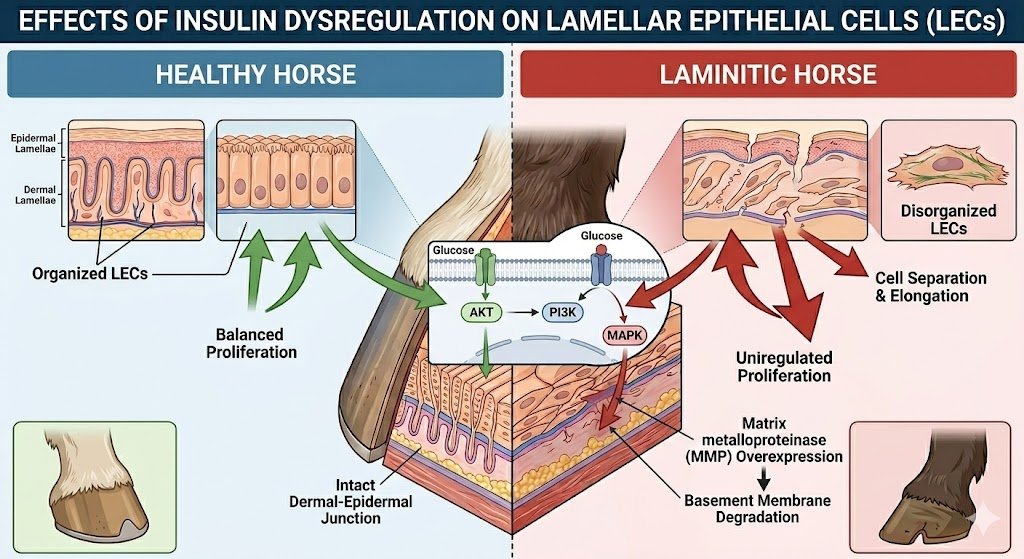
Conclusion
The insulin–IGF-1 receptor theory provides a biologically coherent explanation for endocrinopathic laminitis.
Sustained hyperinsulinemia acts as an upstream endocrine driver that:
- Engages IGF-1R-linked growth signaling
- Promotes lamellar epithelial remodeling
- Reduces structural load tolerance
- Leads to progressive dermo-epidermal weakening
This distinguishes metabolic laminitis from inflammatory forms and integrates:
- Endocrine physiology
- Cellular signaling
- Hoof biomechanics
Understanding this pathway equips veterinarians, farriers, breeders, and advanced owners with a higher-resolution framework for interpreting insulin-related lamellar pathology.
Frequently Asked Questions
Q1: How does insulin overstimulation damage lamellae?
Sustained hyperinsulinemia likely stimulates IGF-1R-linked growth signaling in lamellar epithelial cells, promoting remodeling and weakening attachments.
Q2: Is IGF-1R the main cause?
IGF-1R is a plausible downstream mediator, while insulin dysregulation remains the key upstream driver within a broader metabolic network.
Q3: How is insulin dysregulation diagnosed?
Through resting insulin evaluation and dynamic testing approaches designed to capture post-prandial hyperinsulinemia under standardized conditions.
epithelial stretch.
References
- Asplin, K. E., Sillence, M. N., Pollitt, C. C., & McGowan, C. M. (2007). Induction of laminitis by prolonged hyperinsulinaemia in clinically normal ponies. The Veterinary Journal, 174(3), 530–535.
- de Laat, M. A., McGowan, C. M., Sillence, M. N., & Pollitt, C. C. (2013). A potential role for lamellar insulin-like growth factor-1 receptor in the pathogenesis of hyperinsulinaemic laminitis. The Veterinary Journal, 197(2), 302–306.
- Durham, A. E., Frank, N., McGowan, C. M., Menzies-Gow, N. J., Roelfsema, E., Vervuert, I., Feige, K., & Fey, K. (2019). ECEIM consensus statement on equine metabolic syndrome. Journal of Veterinary Internal Medicine, 33(2), 335–349.
- Equine Endocrinology Group. (2021). Recommendations for the diagnosis and treatment of pituitary pars intermedia dysfunction (PPID).
- McGowan, T. W., Pinchbeck, G. P., & McGowan, C. M. (2013). Prevalence, risk factors and clinical signs predictive for equine pituitary pars intermedia dysfunction in aged horses. Equine Veterinary Journal, 45(1), 74–79.
- Patterson-Kane, J. C., Karikoski, N. P., & McGowan, C. M. (2018). Paradigm shifts in understanding equine laminitis. The Veterinary Journal, 231, 33–40.








