Executive Summary
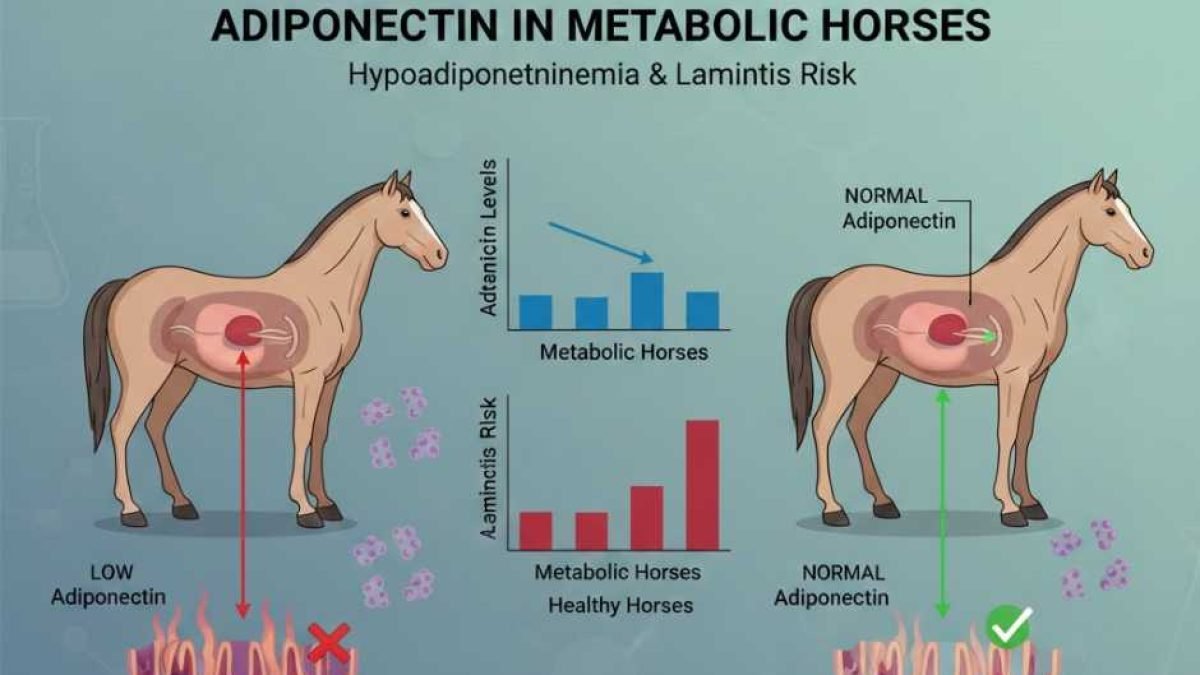
Adiponectin in horses is a fat-derived hormone and one of the most clinically discussed adipokines in equine metabolic disorders. In metabolic horses—particularly those with equine obesity, equine insulin resistance, or broader insulin dysregulation—circulating adiponectin often declines, a state known as hypoadiponectinemia.
Importantly, this decline matters because endocrinopathic laminitis has strong evidence linking risk to hyperinsulinemia. Researchers experimentally induced laminitis through prolonged hyperinsulinemia, clearly demonstrating this connection (Asplin et al., 2007).
Moreover, research supports adiponectin as a relevant adipose tissue biomarker within laminitis prediction frameworks. A cohort study validating adiponectin assays in ponies showed that ponies with a history of endocrinopathic laminitis had lower circulating adiponectin concentrations. The same study found reduced levels in ponies that later developed laminitis (Menzies-Gow et al., 2019).
Therefore, this article explains what low adiponectin means in horses, how hypoadiponectinemia influences insulin regulation, and how these findings integrate into laminitis prevention science.
Introduction
Across modern stables, metabolic horses are increasingly common. Easy keepers gain weight quickly, develop insulin dysregulation, and may show seasonal foot soreness during pasture transition.
However, metabolic instability often develops silently before obvious lameness appears. For this reason, early endocrine markers deserve closer attention.
Within this metabolic landscape, adiponectin has gained attention because it supports insulin sensitivity and healthy glucose–insulin dynamics. When adiponectin drops, metabolic resilience weakens, and endocrinopathic laminitis risk may rise.

What Is Adiponectin?
Equine adiponectin is an endocrine signal produced by adipose tissue and categorized among adipose-derived, fat-derived hormones that shape metabolic regulation. In contrast to leptin, which generally increases with fat mass, adiponectin often decreases in insulin-resistant horses and in equine metabolic syndrome (EMS)-type phenotypes. As a result, adiponectin concentration in horses is often interpreted as a marker of adipose endocrine quality rather than simply the amount of adipose tissue
Adiponectin circulates in multiple molecular forms, including total adiponectin measurement and high-molecular-weight adiponectin fractions. In other mammals, adiponectin receptors (AdipoR1/AdipoR2) influence energy-regulating pathways such as AMP-activated protein kinase signaling, supporting insulin sensitivity and lipid oxidation
Causes of Hypoadiponectinemia
Hypoadiponectinemia typically reflects adipose tissue dysfunction. As adipocytes enlarge with increasing adiposity, endocrine output shifts away from protective signaling.
Consequently, metabolically protective adipokine balance weakens.
Research shows that adiponectin concentrations decrease as adiposity increases. Kearns et al. (2006) demonstrated that adiponectin and leptin correlate with fat mass in horses. Furthermore, Bamford et al. (2016) showed that diet and adiposity alter adipokine concentrations and insulin sensitivity.
Importantly, hypoadiponectinemia often coexists with resting hyperinsulinemia or exaggerated post-prandial insulin responses. When adiponectin signaling declines, insulin responsiveness may decrease. As a result, the pancreas compensates by increasing insulin output over time.
Link With Equine Obesity
Obesity in horses is common at population level. A field study of outdoor-living horses and ponies reported obesity prevalence rising from 27.08% at the end of winter to 35.41% in summer in the same population, with breed as the strongest risk factor (Giles et al., 2014). This seasonal drift is clinically meaningful because pasture sugars and insulin responses often vary with season, influencing equine laminitis risk.
However, obesity and equine metabolic syndrome (EMS) are not synonymous. EMS is defined primarily through insulin dysregulation in horses, not body condition alone (Durham et al., 2019). This is where adipose tissue biomarkers can add nuance: two horses with similar body condition can show different degrees of equine metabolic imbalance, different insulin sensitivity, and different laminitis vulnerability. Adiponectin deficiency in horses, when present, may indicate a more pathologic adipose endocrine function rather than simple weight gain.
Connection to Insulin Dysregulation
Insulin dysregulation is the central risk engine in endocrinopathic laminitis and a defining feature of EMS (Durham et al., 2019). It includes resting hyperinsulinemia, exaggerated post-prandial insulin response, and variable degrees of equine insulin resistance. This framework matters because hyperinsulinemia-induced laminitis has been experimentally demonstrated. Asplin et al. (2007) induced laminitis through prolonged hyperinsulinemia in clinically normal ponies, establishing that insulin dysregulation and lamellar damage can be directly linked.
In this context, adiponectin is relevant because it supports insulin sensitivity. When circulating adiponectin is reduced, insulin signaling efficiency may decline, increasing the likelihood of compensatory hyperinsulinemia. This is why clinicians increasingly frame adiponectin as a metabolic risk marker rather than a diagnostic substitute: it can strengthen interpretation when insulin values are borderline or when phenotype and laboratory results appear discordant.
Importantly, gut-driven effects also matter. The enteroinsular axis in horses refers to gut-to-pancreas signaling that influences insulin secretion in response to dietary carbohydrate effects. This helps explain why some animals show marked insulin spikes after carbohydrate exposure even if fasting markers are modest—an important feature of the glucose–insulin dynamics of metabolic horses.
Laminitis Risk Pathways
Laminitis pathophysiology is increasingly described as a syndrome with distinct upstream drivers. Endocrinopathic laminitis (endocrine-related laminitis) is closely tied to insulin dysregulation and is considered the most common subtype encountered in practice (Menzies-Gow et al., 2015). Mechanistically, insulin-induced laminitis is supported by evidence that insulin can act through IGF-1 receptor laminitis pathways in lamellar epithelial cells, contributing to lamellar pathology in horses (de Laat et al., 2013).

Gut–Metabolic Axis and Dietary Considerations
Within Equine Gut Health & Nutrition Science, the equine gut–metabolic axis is central because dietary carbohydrate effects influence enteroinsular signaling and downstream insulin secretion. Diet change research demonstrates this system-level connection. A study examining diet change in ponies found that adding pasture to a hay diet altered the faecal microbiome and that metabolically healthy animals showed greater microbial stability than insulin-dysregulated ponies (Fitzgerald et al., 2020). This does not prove that the microbiome causes hypoadiponectinemia, but it supports a systems view in which microbiome and insulin regulation are linked.
The practical implication for metabolic physiology is that forage sugars and insulin response are interconnected, particularly during seasonal pasture shifts. This aligns with why non-structural carbohydrates (NSC) and postprandial insulin spikes remain central considerations in laminitis prevention frameworks and why vet-guided monitoring is frequently emphasized for high-risk horses (Durham et al., 2019).
Diagnostic and Monitoring Strategies
Adiponectin is best considered within a multi-marker approach to biomarkers for laminitis rather than as a single diagnostic determinant. EMS biomarkers in practice often center on insulin dysregulation testing, with additional endocrine evaluation when indicated, especially in older horses where PPID and laminitis risk can overlap (Durham et al., 2019). Adiponectin measurement can complement interpretation, particularly when trying to interpret adiponectin levels in horses alongside insulin metrics, phenotype, and prior laminitis history.
Assay methodology matters. The study evaluating measurement approaches for adiponectin in ponies highlights the importance of validated methods. Interpretive caution required when different assays or platforms are used (Menzies-Gow et al., 2019). This is particularly relevant for questions such as total adiponectin vs HMW adiponectin in horses, because the clinical usefulness of fractions depends on assay performance, reference populations, and intended application.
Conclusion
Adiponectin in horses is an informative equine metabolic hormone and an adipose tissue biomarker that contributes to understanding equine metabolic dysfunction. Hypoadiponectinemia is associated with metabolic horses exhibiting insulin dysregulation, equine obesity, and increased endocrinopathic laminitis risk. The evidence linking insulin dysregulation. Laminitis is robust, including experimental induction of insulin-induced laminitis via prolonged hyperinsulinemia. Mechanistic evidence implicating IGF-1 receptor laminitis pathways (de Laat et al., 2013; Baskerville et al., 2018). Population data emphasize the scale of obesity in horses and seasonal drift (Giles et al., 2014), while longitudinal work supports adiponectin as a biomarker associated with laminitis history and subsequent disease development (Menzies-Gow et al., 2019). Taken together, these findings explain why hypoadiponectinemia and laminitis risk continues to receive scientific and clinical attention.
Frequently Asked Questions (FAQs)
Q1: What does low adiponectin mean for my horse’s health?
A: Low adiponectin (hypoadiponectinemia) is generally interpreted as a marker of adipose endocrine dysfunction and reduced metabolic resilience, and it has been associated with endocrinopathic laminitis history and subsequent laminitis development in ponies (Menzies-Gow et al., 2019).
Q2: How hypoadiponectinemia affects insulin regulation?
A: Adiponectin is linked to insulin sensitivity in horses. Reduced adiponectin may align with decreased insulin signaling efficiency and compensatory hyperinsulinemia, which is central to insulin-driven laminitis risk models (Durham et al., 2019).
Q3: Can low adiponectin predict laminitis risk?
A: Evidence supports adiponectin as a risk-associated biomarker. Lower adiponectin has been observed in laminitis-prone ponies and in ponies that later developed laminitis, supporting refined risk stratification rather than a definitive stand-alone prediction (Menzies-Gow et al., 2019).
Call to Action
For horses fitting metabolic risk profiles—such as easy keepers, individuals with prior endocrinopathic laminitis, or older horses with possible PPID overlap—current evidence supports discussing structured metabolic screening with a veterinarian, with particular emphasis on insulin dysregulation assessment and interpretation within the broader equine endocrine system (Durham et al., 2019). Readers seeking additional depth may benefit fromrelated evidence-based discussions on equine obesity, insulin dysregulation, and laminitis prevention pathways.
References
- Asplin, K. E., Sillence, M. N., Pollitt, C. C., & McGowan, C. M. (2007). Induction of laminitis by prolonged hyperinsulinaemia in clinically normal ponies. The Veterinary Journal, 174(3), 530–535.
- Bamford, N. J., Potter, S. J., Harris, P. A., & Bailey, S. R. (2016). Effect of increased adiposity on insulin sensitivity and adipokine concentrations in different equine breeds. The Veterinary Journal, 215, 37–44.
- Baskerville, C. L., Chockalingam, S., Harris, P. A., Elliott, J., & Menzies-Gow, N. J. (2018). The effect of insulin on equine lamellar basal epithelial cells mediated by the insulin-like growth factor-1 receptor. PeerJ, 6, e5945.
- de Laat, M. A., McGowan, C. M., Sillence, M. N., & Pollitt, C. C. (2013). Equine laminitis: Induced by 48 h hyperinsulinaemia in Standardbred horses. Equine Veterinary Journal, 45(4), 419–424.
- de Laat, M. A., Pollitt, C. C., Kyaw-Tanner, M. T., McGowan, C. M., & Sillence, M. N. (2019). Incidence and risk factors for recurrence of endocrinopathic laminitis in horses. Journal of Veterinary Internal Medicine, 33(3), 1473–1482.
- Durham, A. E., Frank, N., McGowan, C. M., Menzies-Gow, N. J., Roelfsema, E., Vervuert, I., Feige, K., & Fey, K. (2019). ECEIM consensus statement on equine metabolic syndrome. Journal of Veterinary Internal Medicine, 33(2), 335–349.
Fitzgerald, D. M., Spence, R. J., Stewart, Z. K., Prentis, P. J., Sillence, M. N., & de Laat, M. A. (2020). The effect of diet change and insulin dysregulation on the faecal microbiome of ponies. Journal of Experimental Biology, 223(7), jeb219154.